ISSUE1752
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Amy Faucard, MLS, Associate Editor has disclosed no relevant financial relationships.
- Discuss the return of ranitidine tablets to the US market.
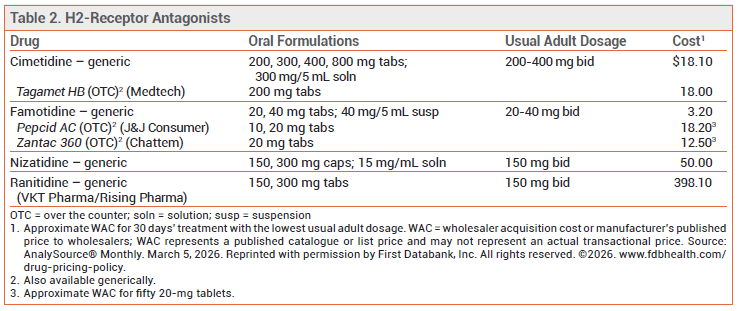
The FDA has approved a new tablet formulation of the H2-receptor antagonist (H2RA) ranitidine from one manufacturer (VKT Pharma/Rising Pharma). It is only available by prescription. In 2020, the FDA requested that all formulations of ranitidine be withdrawn from the market because unacceptable levels of the nitrosamine compound N-nitrosodimethylamine (NDMA), a potentially carcinogenic contaminant, had been detected in ranitidine samples.
NDMA — NDMA can be found in drinking water (as a byproduct of water disinfection) and in some processed foods. It has been found in many drugs, including nizatidine, which is structurally related to ranitidine. High levels of NDMA have been shown to induce tumors in multiple organs in animals; it is classified as a probable human carcinogen.
FDA testing found that NDMA levels in the original ranitidine formulations increased over time and when the drug was stored at high temperatures. Approval of the reformulated tablets was based on changes in the manufacturing process that improve the stability of the drug, minimizing formation of NDMA during its shelf life.
AVAILABILITY — Prior to removal, ranitidine was available over the counter (OTC) and by prescription (Zantac, and generics).1 The active ingredient in OTC Zantac 360 is now the H2RA famotidine (see Table 2).
The new formulation is available by prescription in bottles of 30 tablets containing either 150 mg or 300 mg of ranitidine. The bottles should be stored at 15-30°C (59-86°F) in a dry place and protected from light. The updated label states that the tablets must be dispensed in the original container. The label also includes storage and handling instructions for patients (see Table 1).
FDA GUIDANCE — The FDA recently issued guidance on acceptable intake (AI) limits for NDMA and some other nitrosamine impurities in drugs. The AI represents the level at or below which the impurity would not be considered a safety concern.2 The updated ranitidine label states that the formulation meets FDA-approved specifications for nitrosamine impurities.